Omics
Nowadays, the so-called "omics" technologies are among the most important pillars in the life sciences. Through bioanalytical high-throughput methods, large amounts of data from biological processes can be obtained in a short time, leading, among other things, to a better understanding of intra- and intercellular processes. Besides the classical life sciences such as biology, biochemistry, medicine, and pharmacology, fields like biotechnology, food technology, nutrition, and environmental sciences are increasingly benefiting from these technologies.
As an example of metabolomics, a heatmap is presented here showing the relative changes of selected metabolites in two life cycle stages of the coccolithophore Emiliania huxleyi under different culture conditions.
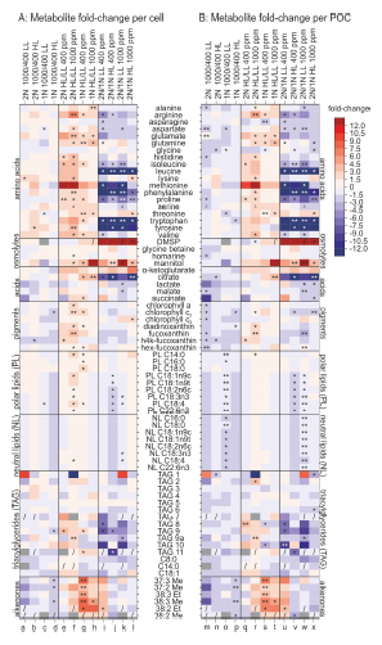
Heidenreich, E et al .(2019) Ocean acidification has little effect on the biochemical composition of the coccolithophore Emiliania huxleyi; Plos ONE; 14 (7)
Proteomics
HDAC8 belongs to the enzyme family of zinc-dependent histone deacetylases (HDAC) and is known to be regulated by a redox switch. The enzyme plays an important role in various cancers, particularly in childhood neuroblastoma. This study provides a deep insight into the regulation of HDAC8 enzyme activity through disulfide bonds induced by PD-404,182, a covalent inhibitor of HDAC8.
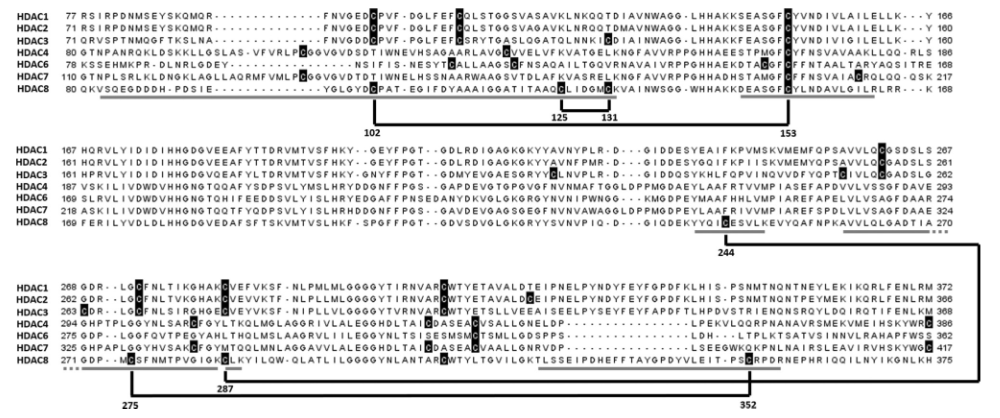
Structural alignment of various HDAC proteins. Cysteines are marked in black, and disulfide bridges are represented by black lines. Theoretical peptide fragments are indicated by gray bars below the alignment.
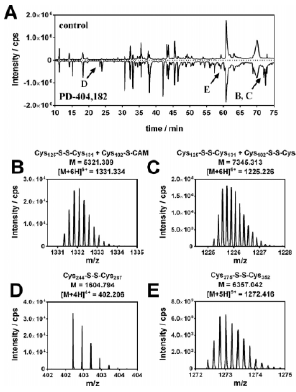
HPLC-MS/MS analysis of disulfide-linked, tryptic HDAC8 peptides. A) HPLC chromatogram of the untreated and PD-404,182 treated HDAC8 samples. B) Mass spectra for the Cys125-S-S-Cys131@S-CAM, C) Cys125-S-S-Cys131 + Cys102-S-S-Cys153, D) Cys244-S-S-Cys287, E) Cys275-S-S-Cys352 peptide fragment.
