Bioprocess-and Reactor Development

The Bioprocess and Reactor Development group advances electrochemical reactor engineering at the interface of process engineering, electrochemistry, and biotechnology, with the goal of translating fundamental insights into scalable, industry-ready technologies. Our research combines reactor design, mechanistic understanding, and process intensification to enable efficient and controllable (bio)electrochemical conversions. In recent years, we have placed a strong emphasis on the development and optimization of particle-electrode reactors. These systems offer exceptionally large electrochemically active surface areas and provide excellent heat and mass transfer, making them highly attractive for high-throughput and high-performance electrochemical and electro-biotechnological processes. We investigate reactor hydrodynamics, transport phenomena, and electrode architectures, and tailor operating strategies to maximize conversion, selectivity, and energy efficiency. Building on successful projects across (bio)process engineering, we have designed scalable reactor concepts and translated them with industrial partners through technology transfer, patenting, and commercialization. Our expertise spans reactor construction and multiphysics simulation, systematic scale-up from lab to pilot scale, and the integration of reliable real-time process monitoring to support data-driven operation, reproducibility, and process control.
Team

from left to right: M.Sc. Yang-Yang Tim Wang PhD-student; M.Sc. Sebastian Castro PhD-Student; Middle: Dr. Andre Tschöpe Head of Process and Reactor Technology; M.Sc. Dominik Moosmann PhD-Student; M.Sc. Michael Abt PhD-Student
List of staff members
Group Leader

+49 721 608-22986
andre tschoepe ∂does-not-exist.kit edu
Geb. 330 / R. 238
PhD-Students

+49 721 608-23794
michael abt ∂does-not-exist.kit edu
Geb. 330 / R. 237

+49 721 608-23794
yang-yang wan ∂does-not-exist.kit edu
Geb. 330 / R. 237

+49 721 608-23794
dominik moosmann ∂does-not-exist.kit edu
Geb. 330 / R. 237

+49 721 608-23794
sebastian castro ∂does-not-exist.kit edu
Geb. 330 / R. 237
Latest Research Topics
|
M.Sc. Dominik Moosmann |
Process intensification of electrochemical particle reactors for CO₂ conversion into biotechnologically usable carbon sources |
|
|
|
|
M.Sc. Sebastian Castro |
Electrochemical Cu-catalysed Particle-bed Reactor for CO2 to Acetate conversion |
|
|
|
|
M.Sc. Yang-Yang Tim Wang |
Electrochemical fluidized bed reactors for electro-enzymatic syntheses including gaseous phase |
|
|
|
|
M.Sc. Michael Abt |
Magnetically enhanced electrochemical fluidized bed reactors for electro-enzymatic syntheses including gaseous phase |
A current list of possible thesis topics can be found here
Research Topic
Description
CO2 is to be made usable as a resource, for example in the form of C2 molecules as platform chemicals or as a nutrient basis for microbial conversions, in order to re-enter the value chain. To this end, a reactor is to be developed in which CO2 dissolved under overpressure is converted to acetate at a particle electrode. The electrode surface area, which is greatly increased by the particles, is intended to raise the production rate and efficiency to a level suitable for application and allow scaling to an industrially usable scale. The selectivity of the copper particles used will be improved by adding a suitable catalyst in order to achieve higher acetate concentrations.
The focus of this project is on developing a suitable electrode and optimizing the catalyst. The necessary experiments will be carried out in specially designed and 3D-printed electrochemical cells on a mL scale. The test setup and parts of the analysis are to be automated and linked to enable parallel testing. The catalysts and catalyst variants to be investigated must be synthesized and characterized, and the resulting C-products analyzed.
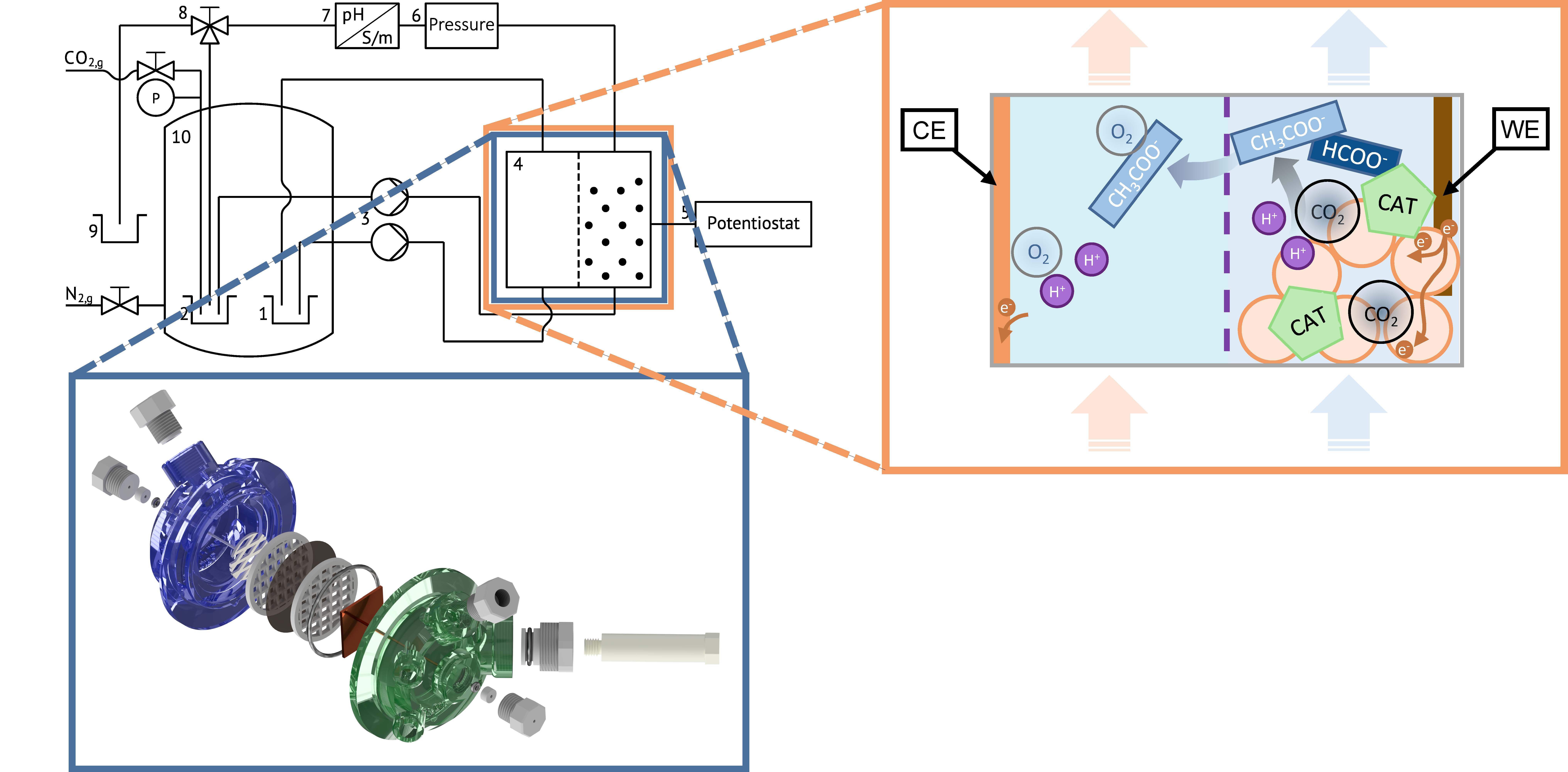
Fig. 1: Schematic of the experimental high-pressure setup. Blue: 3D render of the electrochemical cell with a three-electrode setup consisting of Counter Electrode (CE), Working Electrode (WE, particles not pictured) and Reference Electrode (RE). Orange: Working principle of the catalyst assisted CO2 reduction reaction inside the electrochemical cell.
Person in Charge

+49 721 608-23794
sebastian castro ∂does-not-exist.kit edu
Geb. 330 / R 237
Research Topic
Magnetically enhanced electrochemical fluidized bed reactors for electro-enzymatic syntheses including gaseous phase
Description
Traditional processes for chemical synthesis use fossil resources and cause emissions detrimental to the climate. An innovative and green alternative presents electro-biosynthesis that combines electrochemistry with biotechnology. The concept of electro-biosynthesis is to use electricity generated by renewable energy, green catalysts (e.g. enzymes) and renewable resources in order to produce chemicals “carbon neutral”. To achieve competitive electro-biosyntheses, we aim to reach high space-time yields which require electro-bio reactors with high volume specific electrode surfaces.
The main objective of the research project is the development, characterization and exemplary application of a magnetically enhanced electrochemical fluidized bed reactor for electro-enzymatic synthesis. The reactor will realize a novel variant of a particle based electrochemical reactor combining the advantages of the very high volume specific electrode surface of particle electrodes with the good mixing and mass transfer properties of e.g. bubble column reactors. The beneficial combination results from the fluidization of the particle electrode while at the same time keeping a conductance due to a magnetically induced chaining of the particles. In comparison to reactors applying numbering up of flat electrodes, the scale-up of a particle electrode is simple and economic.
The efficiency of the novel reactor concept for electro-enzymatic synthesis will be demonstrated by the in-situ generation of hydrogen peroxide which is required for oxyfunctionalization reactions using peroxygenases. The whole project will be accompanied by a close feedback between experimental results and multiphysics modelling approaches. The reactor prototypes will be produced via 3D printing technology.
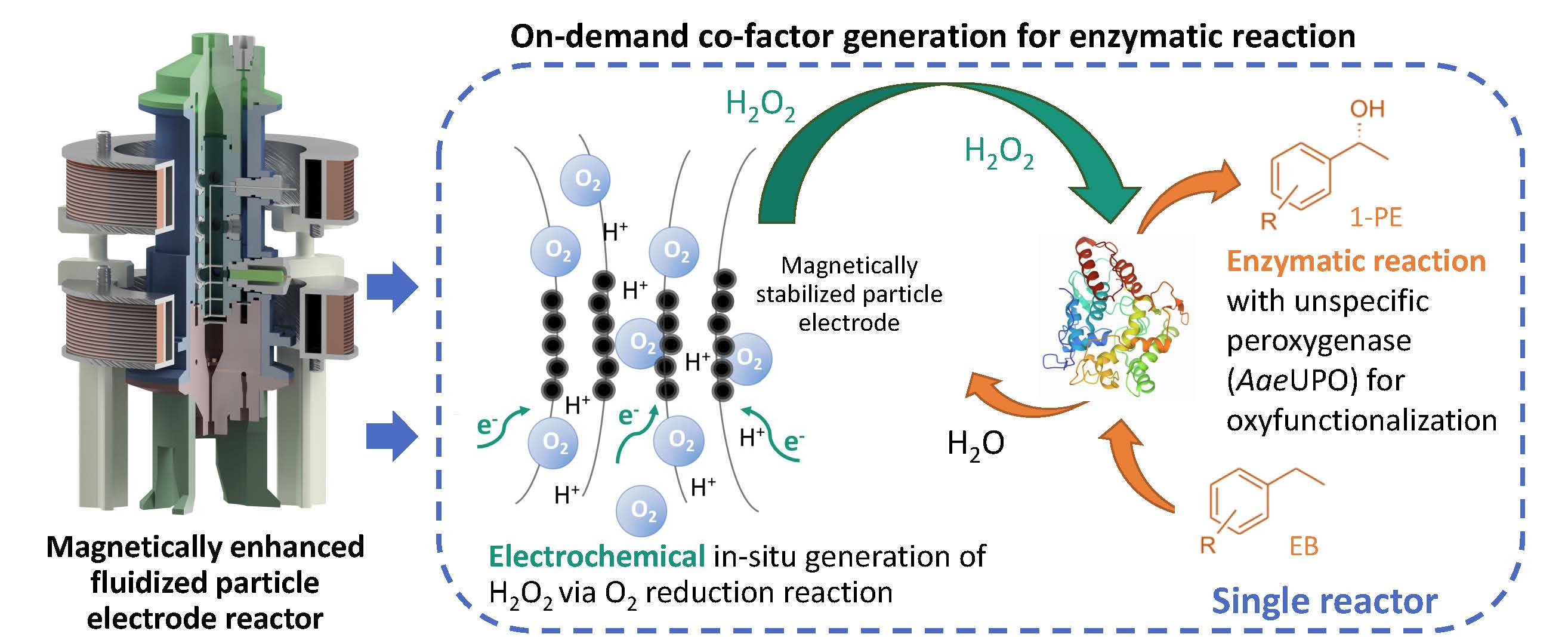
Figure 1: Schematic illustration of a magnetically enhanced fluidized bed electrode operated with three phases: magnetizable particle electrode (black particles), liquid phase and gas phase (O2 bubbles). In the reactor we generate hydrogen peroxide in-situ via a particle electrode by oxygen reduction reaction. The enzyme, a peroxygenase, uses hydrogen peroxide as
co-substrate for the oxyfunctionalization of ethylbenzene (EB) to 1-phenethyl alcohol (1-PE).
Person in Charge

+49 721 608-23794
michael abt ∂does-not-exist.kit edu
Geb. 330 / R. 237
Research Topic
Process intensification of electrochemical particle reactors for CO₂ conversion into carbon sources usable in biotechnology
Description
Conventional synthesis routes for valuable fine chemicals typically rely on fossil feedstocks and are associated with climate-relevant emissions. A promising alternative is to first reduce dissolved CO₂ electrochemically, using electricity from renewable sources, into carbon-based platform chemicals such as acetate and formate, and then upgrade these intermediates microbially to fine chemicals. New processes and reactors in electrobiotechnology, the combination of electrochemistry and biotechnology, thus open an attractive pathway towards sustainable production in the chemical industry. Despite its potential, industrial deployment of this technology is still in its infancy. Key challenges include long-term stability, scalability, and seamless integration into existing process chains.
The goal of this research project is to develop a novel electrochemical platform technology for the industrial production of acetate from CO₂. At its core is a modular, scalable high-pressure particle-electrode reactor that, for the first time, exploits the potential of three-dimensional particle electrodes for CO₂ reduction. Compared with conventional planar electrodes, this system offers a greatly enlarged active surface area, improved mass transfer, and higher current (Faradaic) efficiency. All essential prerequisites for economic operation up to the industrial scale. The project is structured into four consecutive work packages: from proof-of-concept at laboratory scale, through optimization of reactor design, operating parameters, and catalysts, to scale-up towards a litre-scale reactor for subsequent integration into biotechnological production processes. The long-term objective is to establish a CO₂-neutral synthesis process that replaces fossil carbon sources and contributes to the decarbonization of industrial value chains.
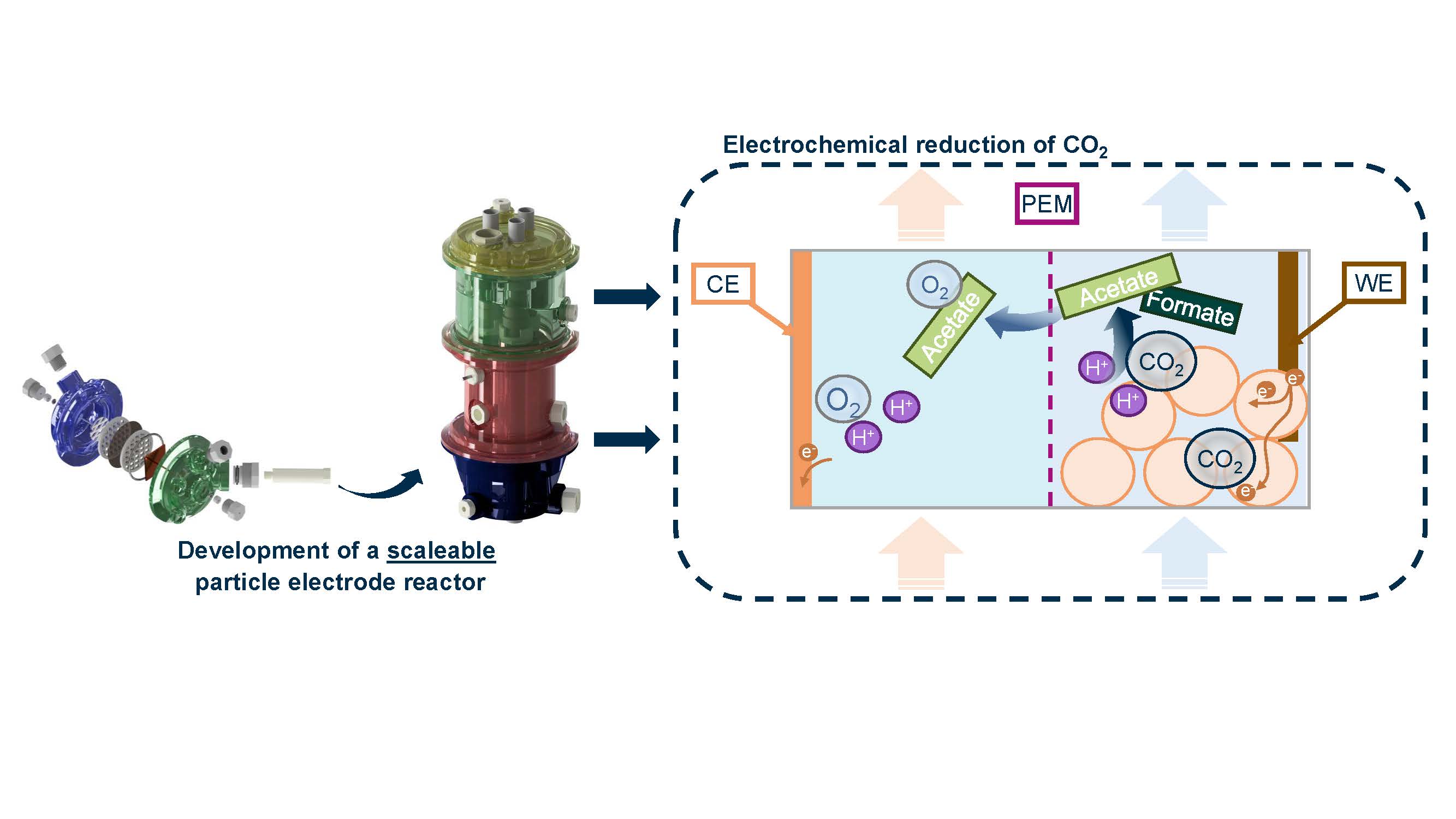
Figure 1: Schematic representation of the scaleable particle electrode reactor. The working electrode chamber and the counter electrode chamber are separated by a proton exchange membrane (PEM). A current distributor connected to the power source outside the chamber is used to polarize the particles. The CO₂ is dissolved in the working electrode chamber and is reduced to acetate or formate at the particle surface (working electrode, WE). The acetate can migrate through the PEM into the counter electrode chamber and is thus separated from the formate. Water electrolysis takes place at the counter electrode (CE) to provide electrons
Person in Charge

+49 721 608-23794
dominik moosmann ∂does-not-exist.kit edu
Geb. 330 / R. 237
Research Topic
Electrochemical fluidized bed reactors for electroenzymatic syntheses including gaseous phases – Modeling, scale-up and in-line process monitoring
Description
New technologies are needed to achieve the goals for sustainable development. Electrobiotechnology offers many opportunities here by combining the advantages of electrochemistry and biotechnology. However, to date, electrobiotechnological processes have mainly been carried out on a laboratory scale. Scalable reactor systems can change this and thus enable the transfer from laboratory to industrial scale. Research priorities in the further development of laboratory processes are, for example, upscaling, integration into existing plant systems, optimization of the biocatalyst and the electrode material and the architecture of the reactors.
Of particular interest are scalable reactor systems using particle electrodes. Particle electrodes are among the so-called 3D electrodes and offer a promising approach for scaling. They consist of a bed of conductive electrode particles (e.g. graphite) and have a large electrode surface-to-solution ratio. The advantage of this is that even low reactant concentrations are sufficient for the process. Particle electrodes are classically used as fixed-bed or fluidized-bed electrodes. Fluidized bed electrodes have the additional advantage that they have very good heat and mass transfer properties.
The aim of the research project is the development, characterization and exemplary application of a scalable fluidized bed reactor for electrochemical synthesis. The reactor is based on a reactor system previously developed in House, which will be scaled up and converted into a three-phase system. The system is to be characterized and optimized using electroenzymatic model reactions. For the reaction, electrochemically generated hydrogen peroxide is used as a co-substrate for the non-specific peroxygenase, which catalyzes a hydroxylation reaction. In addition, the process understanding is to be deepened by integrating online analytics and limitations are to be identified through modeling. 3D printing is used as a method for developing the reactor prototypes.
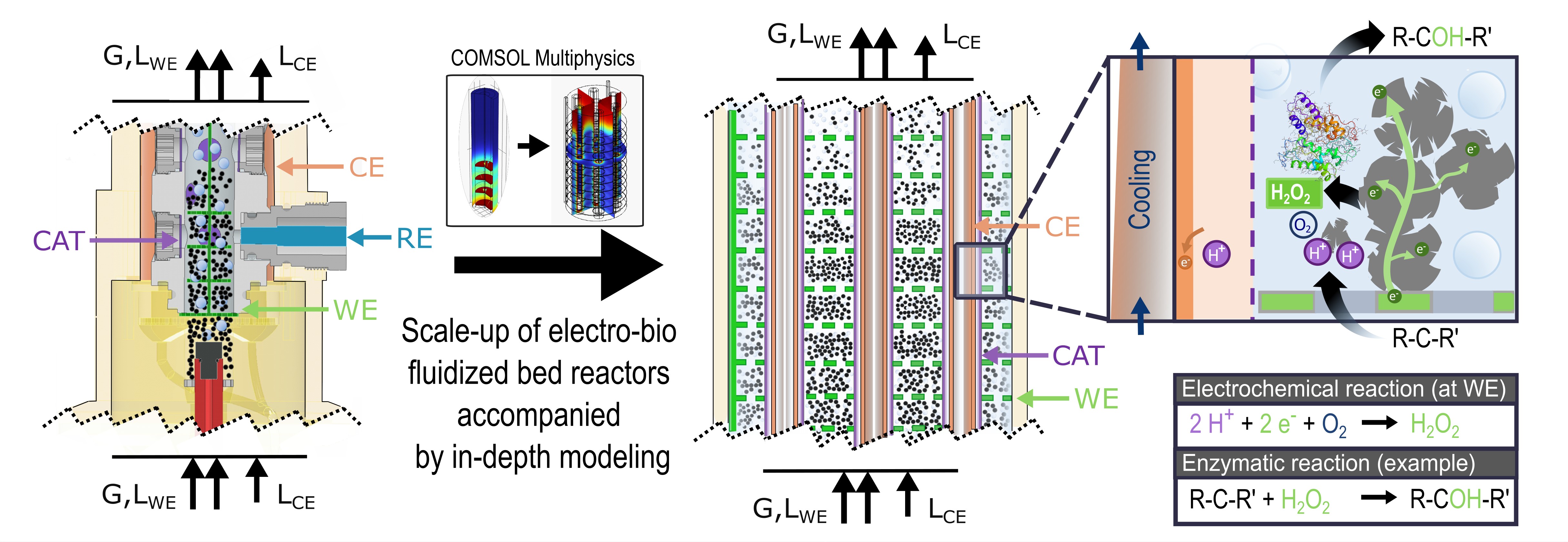
Figure 1: Schematic illustration of the reactor principle. The reactor consists of a particle working electrode which is separated from the platinized titanium rods counter electrode by a cation exchange membrane. Hydrogenperoxide (H2O2) is generated in situ on the particle surface. The required hydrogen is supplied from the CEC. To supply oxygen a sparger is integrated in the WEC. H2O2 is used as co-substrate for the enzymatic hydroxylation. Furthermore, the modeling supported scale up from mL to simple L scale is illustrated.
Person in Charge

+49 721 608-23794
Yang-yang wang ∂does-not-exist.kit edu
Geb. 330 / R. 237
